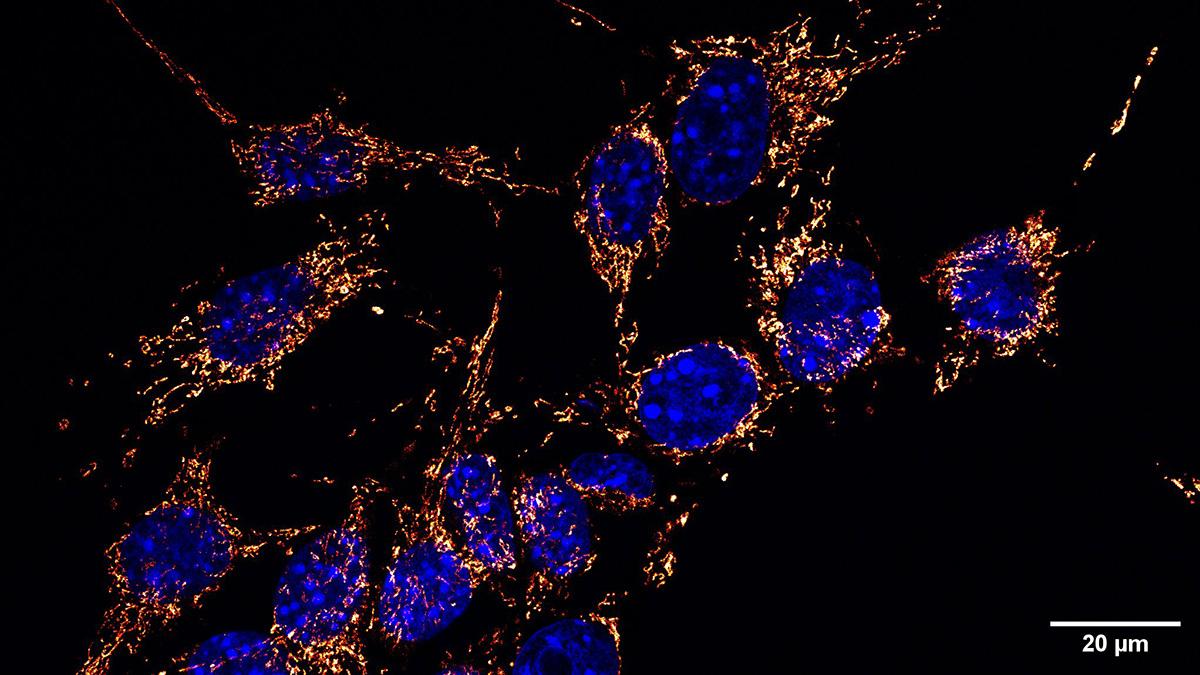
Mitochondrial Transport of Methylated Amino Acids
A multi-institute research group led by Thomas MacVicar at the Cancer Research UK Scotland Institute, Glasgow, found that a specific transporter called SLC25A45 is required for the transport of methylated amino acids across the inner mitochondrial membrane and for carnitine synthesis. This study was published as a recent paper in Molecular Cell 1.
For cellular metabolism to function effectively, metabolites must be exchanged between the mitochondria and the cytosol. Crossing the inner mitochondrial membrane requires the action of transporter proteins. Disruptions to these transporters can lead to disease. The most common transporters are the members of the solute carrier (SLC) 25 family. However, the substrates for some transporters are unknown. Among these “orphan transporters” was solute carrier (SLC) 25A45.
Mitochondria are deeply involved in amino acid metabolism, but the researchers wondered how they deal with methylated amino acids. Disruptions to the systems regulating methylated amino acid homeostasis are associated with heart and kidney diseases, and some are associated with tumors. The team found that a specific transporter, SLC25A45, binds to dimethylarginine and trimethyllysine, but not to the unmethylated version of these amino acids.
Identifying the substrate for orphan transporters is valuable knowledge. SLC25A45 is particularly important for its roles in the transport of methylated amino acids and carnitine biosynthesis. This research also suggests some new possible therapeutic strategies.
A conversation with Dr. MacVicar:
MitoWorld: Can you describe the directions of your research to further the findings of this paper?
MacVicar: Now we know that methylated amino acids enter mitochondria via SLC25A45, we are keen to understand how this pathway impacts cancer progression. The Birsoy and Kajimura labs have shown in mice that mitochondrial uptake of trimethyllysine, and subsequent biosynthesis of carnitine, is important for physiological responses that depend on fatty acid oxidation 2,3. We are employing cancer mouse models to explore how mitochondrial metabolism of trimethyllysine and other methylated amino acids impact tumour growth and survival.
MitoWorld: Do you have any plans to tackle other orphan transporters?
MacVicar: Orphan solute carriers are crucial pieces missing from the mitochondrial metabolism jigsaw. With continued collaborative and creative research, I’m optimistic that each member of the SLC25 family will be deorphanised within 5 years. It will be a challenge though, in part because some orphan transporters appear to have specialised tissue-specific roles. As fun as the SLC25A45 project was, we don’t currently have plans to take on any more orphans ourselves. We have much to learn about the regulation of SLC25 protein biogenesis and activity, and I also think it’s important to study non-SLC25 mitochondrial metabolite transporters. This includes interesting metabolite transport proteins that appear to be dual-localised between mitochondria and other cell membranes. I’m excited to see what comes next from this dynamic field.
MitoWorld: Methylation is a common post-translational modification. Can you speculate on why the mitochondria have become specialized in dealing with some of these?
MacVicar: here may be several advantages of compartmentalising methylated amino acid metabolism. As mentioned, cells can control carnitine biosynthesis by regulating mitochondrial import and hydroxylation of trimethyllysine in the matrix. Whereas mitochondrial sequestration of dimethylarginine perhaps controls nitric oxide synthesis, which is inhibited by cytosolic accumulation of dimethylarginine. By importing methylated amino acids, mitochondria may somehow play a role in sensing the downstream products of methionine metabolism and protein catabolism. Of course, this remains very speculative for now!
MitoWorld: We always wonder how you became interested in mitochondria. Can you expand on that?
MacVicar: I was hooked on mitochondria after some live-cell imaging experiments at the beginning of my PhD. I was surprised and fascinated by the interconnected and dynamic behavior of the mitochondrial network, which was not something I’d gathered from textbooks.
References
- Dias MM, King MS, Shokry E, Lilla S, Paul N, Thomason P, Zanivan S, Sumpton D, Kunji ER, MacVicar T (2025) SLC25A45 is required for mitochondrial uptake of methylated amino acids and de novo carnitine biosynthesis. Molecular Cell 85: 4093–4104.
https://www.cell.com/molecular-cell/pdfExtended/S1097-2765(25)00703-8
- Khan A, Yen FS, Unlu G, DelGaudio NL, Erdal R, Xiao M, Wangdu K, Cho K, Gamazon ER, Patti GJ, Birsoy K (2025) Machine-learning-guided discovery of SLC25A45 as a mediator of mitochondrial methylated amino acid import and carnitine synthesis. Cell Metabolism 37: 2220-32.
https://doi.org:10.1016/j.cmet.2025.09.015
- Auger C, Nishida H, Yuan B, Silva GM, Fujimoto M, Li M, Katoh D, Wang D, Granath-Panelo M, Shin J, Witte R (2026) Mitochondrial control of fuel switching via carnitine biosynthesis. Science 391: eady5532.