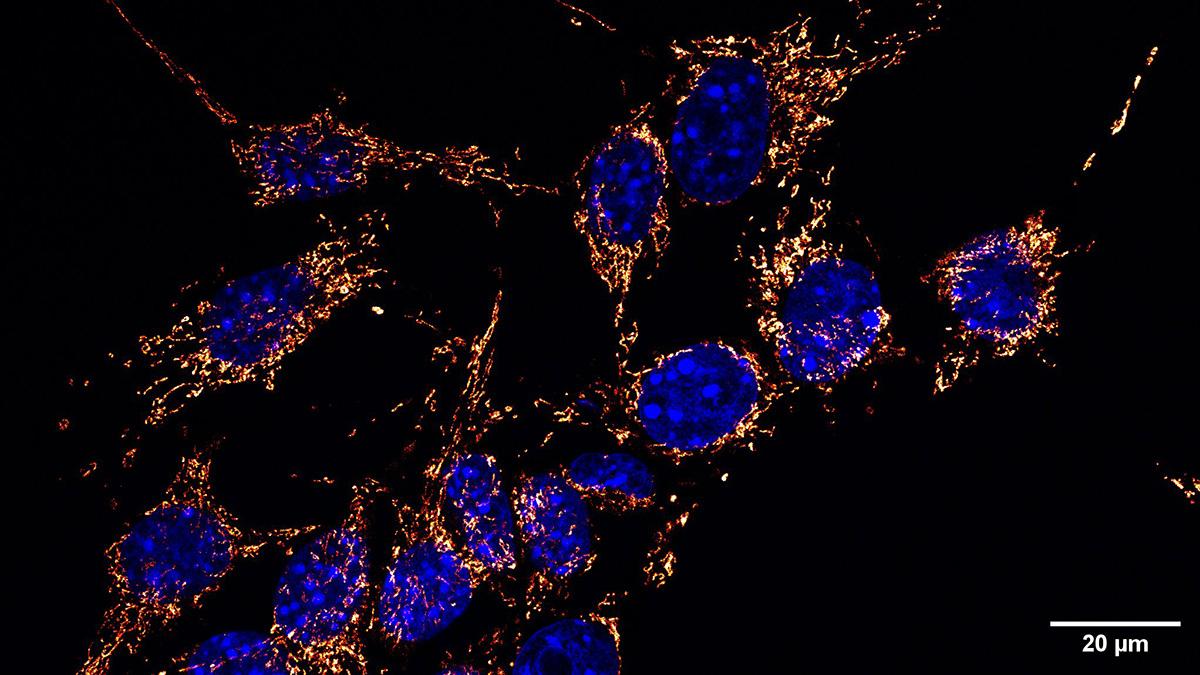
Figure | Mitochondrial network in tumor cells. Representative immunofluorescence images from two optical planes of a Z-stack showing the mitochondrial network in tumor cells. Mitochondria are shown in orange/red and nuclei in blue. Scale bar: 20 µm.
Melanoma Cells Release Abnormal Mitochondria That Might Be Valuable Biomarkers
Melanoma cells release abnormal mitochondria into the extracellular compartment. This novel mitochondrial quality-control mechanism points to possible biomarkers for the disease. The results of the study were recently published in a paper in Cancer Letters and led by Francisca Alcayaga-Miranda.
Melanoma cells experience significant levels of oxidative stress. While they depend on glycolysis, they still need mitochondria for other cell functions. Other studies had shown that damaged mitochondria are expelled from a stressed cell. However, it was not known if this process occurred with melanoma cells and how it was accomplished.
The research team sought to clarify this process. Using electron microscopy, they examined mitochondria released from normal and melanoma cells from mice. All cell lines released damaged mitochondria. The differences were found in the amounts of mitochondria released and the routes. The mitochondria were released through a non-vesicular route. Furthermore, the expelled mitochondria lacked cristae and had multiple other indications of loss. Complementary analyses showed that melanoma cells do not degrade mitochondria through canonical mitophagy in conditions of oxidative stress. Instead, melanoma cells upregulate mitochondrial release to the extracellular medium. With this data, the team concluded that the melanoma cells release mitochondria as an alternative mitochondrial quality control mechanism.
Interestingly, the mitochondria expelled from the melanoma cells were detected in the tumor microenvironment and plasma of the mice. The levels of those mitochondria correlated with the tumor burden. In agreement with these findings, more total extracellular mitochondria were also detected in the circulation of melanoma patients, supporting the translational relevance of this phenomenon.
The study revealed a novel mechanism for releasing dysfunctional mitochondria. It also points to new strategies for non-invasive biomarkers and therapies.
A Statement of Significance from Dr. Alcayaga-Miranda:
This study expands the conceptual framework of mitochondrial quality control in cancer by showing that melanoma cells can externalize structurally and functionally altered mitochondria into the tumor microenvironment and circulation. Rather than being confined to intracellular degradation pathways, mitochondrial dysfunction in melanoma may generate extracellular signals that reflect tumor-associated stress, mitochondrial quality-control imbalance, tumor burden, and systemic disease progression. These findings open two important horizons: first, the development of minimally invasive biomarkers based on circulating extracellular mitochondria, and second, the need to define whether tumor-derived mitochondrial material is only a consequence of mitochondrial stress or also an active mediator of immune modulation and metastatic niche formation. Thus, this work provides a foundation for future mechanistic and translational studies aimed at understanding extracellular mitochondria as both measurable indicators and potential functional players in melanoma progression.
A Conversation with Dr. Alcayaga-Miranda:
MitoWorld. This is rich study filled with experimental detail. Can you give us an idea of what you are considering to follow up on this research?
Dr. Alcayaga-Miranda: Our next step is to determine whether extracellular mitochondria released by melanoma cells are merely a consequence of mitochondrial stress or whether they actively contribute to tumor progression. We are particularly interested in understanding how this mitochondrial material interacts with the tumor microenvironment, especially immune cells, and whether circulating extracellular mitochondria can be further developed as minimally invasive indicators of tumor burden and disease progression.
MitoWorld. It is interesting that multiple mechanisms are available to expel mitochondria. Do you have any thoughts on why these exist?
Dr. Alcayaga-Miranda: Cells likely rely on more than one mechanism to eliminate or externalize mitochondria because mitochondrial quality control is highly context-dependent. Depending on the intensity of cellular stress and the capacity of intracellular degradation pathways, damaged mitochondria may either be degraded within the cell or exported through alternative routes. In melanoma, our findings suggest that extracellular mitochondrial release may represent an additional layer of mitochondrial quality control, particularly when mitochondrial stress is sustained or canonical degradation pathways are insufficient.
MitoWorld. You indicate that melanoma release mitochondria into the bloodstream. It is amazing that so many mitochondria are circulating. Do you have any estimates of that or of how long they circulate before being completely eliminated?
Dr. Alcayaga-Miranda: At this stage, we do not yet have precise estimates of how long extracellular mitochondria remain in circulation. This is an important open question. Their persistence is likely influenced by structural integrity, association with vesicular or non-vesicular compartments, recognition by phagocytic cells, and clearance by organs involved in filtering circulating particles. Future kinetic studies will be necessary to define their half-life, clearance routes, and whether these parameters change during tumor progression.
MitoWorld. There have been other reports that mitochondria are transferred from tumor cells to normal cells, such as immune cells. Did you see any evidence of this in your studies?
Dr. Alcayaga-Miranda: In this study, we focused primarily on demonstrating that melanoma cells release dysfunctional mitochondria into the tumor microenvironment and circulation. We did not directly evaluate mitochondrial transfer to immune cells as a central endpoint. However, the detection of tumor-derived mitochondrial material in the tumor microenvironment raises the possibility that these structures may interact with stromal or immune cells. This is one of the directions we are now actively exploring, particularly in relation to how tumor-derived mitochondria may influence antitumor immune function.
MitoWorld. Do you have any plans to pursue the development of biomarkers?
Dr. Alcayaga-Miranda: Yes. One of the translational horizons of this work is to determine whether circulating extracellular mitochondria can be developed as minimally invasive biomarkers. Before clinical implementation, several steps are required, including analytical standardization, rigorous control of pre-analytical variables during blood processing, validation in larger patient cohorts, and comparison with established clinical parameters. Our current data provide a strong rationale for this direction, but biomarker development will require systematic validation.
MitoWorld. We are always interested in what sparked your interest in mitochondria. Can you tell us?
Dr. Alcayaga-Miranda: My interest in mitochondria began with the idea that they are not only intracellular powerhouses, but also dynamic signaling organelles capable of shaping cell behavior and intercellular communication. In cancer, this is particularly fascinating because tumor cells continuously adapt their metabolism under stress. Understanding how mitochondria move beyond the cell may reveal new dimensions of tumor biology and open unexpected translational opportunities.
Reference
Georges-Calderón N, Fuentes C, Hidalgo Y, Grunenwald F, Corrales-Bermúdez J, Figueroa-Valdés AI, Ramirez-Pereira M, Arriagada G, Bustos FJ, Ahumada-Marchant C, Lopez M, Alcayaga-Miranda F (2026) Melanoma cells release dysfunctional mitochondria to the tumor microenvironment and circulation in association with tumor progression. Cancer Letters 647: 218457.
https://www.sciencedirect.com/science/article/pii/S030438352600220X?via%3Dihub