Glutathione, Mitochondria and Breast Cancer Metastasis
Cancer cells need changes to metabolism to supply energy, especially during metastasis. In a recent paper in Cancer Discovery 1, a research team led by Kivanç Birsoy compared the metabolites of normal and breast cancer cells. They found that metastasis of the cancer cells relied on mitochondrial glutathione. This discovery provides insights into the process of metastasis and implicates glutathione as a potential therapeutic target for breast cancer.
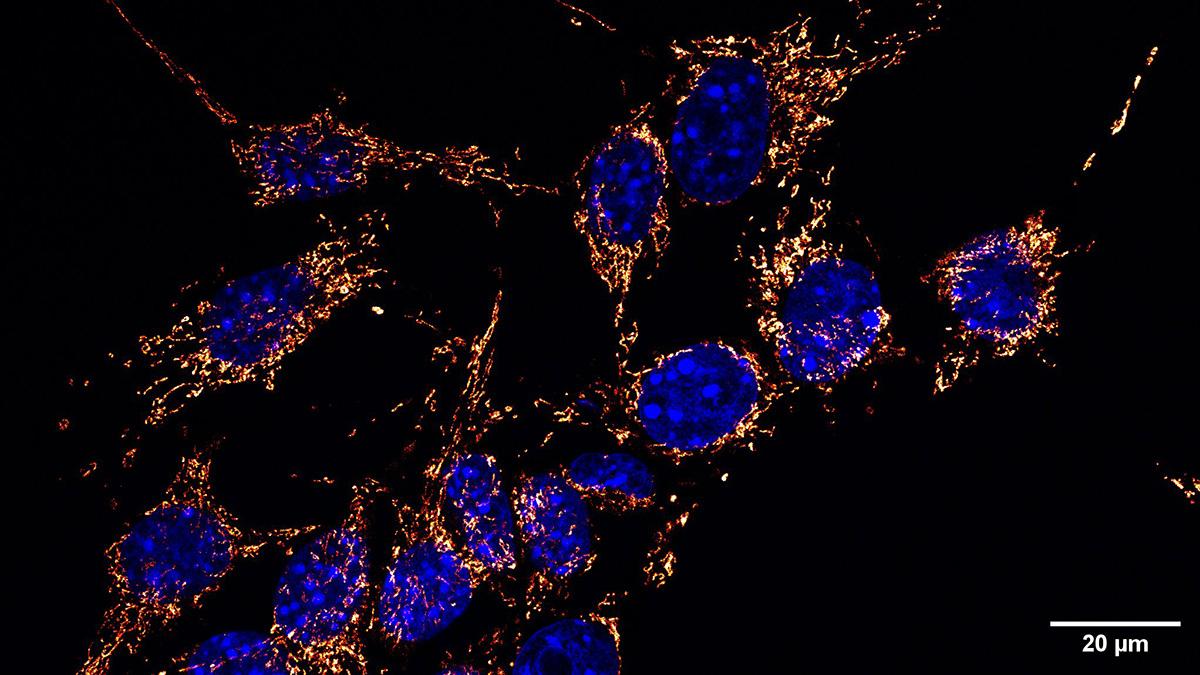
The team compared the primary and metastatic breast cancer cells. This metabolomics study revealed accumulation of more mitochondrial glutathione in metastatic cells. Further analysis identified increased expression of SLC25A39. In previous work, the Birsoy group showed that this mitochondrial membrane carrier regulates glutathione transport into mitochondria.2 SLC25A39 is required for metastases, but its loss has no effect on primary tumors. Interestingly, the team found that the stress-induced transcription factor ATF4 could rescue SLC25A39-deficient metastatic cells. This observation links SLC25A39 to activation of ATF4.
This work provides new insights into the processes of metastasis. It defines a new role for glutathione beyond its well-know role antioxidant role. Finally, it suggests that glutathione, SLKC25A39, and ATF4 as possible therapeutic targets for breast cancer.
A Conversation with Dr. Kıvanç Birsov
MitoWorld: What is the next step in continuing your research?
Dr. Birsov: We’ll dissect how mitochondrial glutathione (mtGSH) is required for ATF4 activation. Specifically, we aim to identify the ISR node that senses mtGSH loss (e.g., GCN2 vs. PERK) and map the molecular steps that couple mitochondrial redox to ATF4. In parallel, we’ll define the precise stresses in the lung that drive this dependency using in vivo stress reporters and targeted perturbations. More broadly, our data suggest there are additional “hidden” metabolites that govern metastatic fitness. With our organelle-resolved metabolomics and genetic tools, we’re poised to systematically uncover these metabolites and their transport pathways.
MitoWorld: Can you speculate on the mechanism by which glutathione enhances metastasis? Do you think the effect is felt by making cells lose their connection to other cells or by helping in the actual colonization at a distant site?
Dr. Birsov: It’s a bit more complex. Sean Morrison’s elegant work has shown that glutathione is essential for tumor cell survival in circulation by buffering oxidative stress. Our findings extend this concept but emphasize that the compartmentalized functions of redox metabolites can differ. In our case, mitochondrial glutathione seems to be particularly important during the colonization phase rather than dissemination. We think its role may go beyond antioxidant defense—potentially involving redox-dependent signaling that supports metabolic adaptation and growth at the metastatic site. But this is still an open question and a major focus of our ongoing work.
MitoWorld: Lung and liver are common sites of metastases. Might this connection be generalized for other forms of metastasis or to other organs?
Dr. Birsov: The environments of metastatic sites are vastly different; each imposes unique metabolic stresses and nutrient availabilities. The lung, for instance, is highly oxidative, and the liver can be hypoxic. Because of these differences, I expect distinct metabolic mechanisms to underlie colonization in each organ. Mitochondrial glutathione may be critical in some contexts, but other metabolites and pathways likely take over in different tissues.
MitoWorld: ATF4 seems to be a two-edged sword. It is activated in stress situations, but aids metastasis. Do you have any thoughts about these strange multiple activities?
Dr. Birsov: That’s exactly what makes ATF4 so fascinating. It’s part of the integrated stress response, normally activated to restore homeostasis when cells face nutrient or redox stress. Cancer cells hijack this pathway—what’s meant to be a transient adaptation becomes chronically active, allowing them to survive and grow in hostile environments. In our system, ATF4 activation seems to compensate for the loss of mitochondrial redox balance, effectively turning a stress response into a metastatic advantage. It’s a perfect example of how tumors repurpose normal adaptive programs for malignant ends.
MitoWorld: Do you have any thoughts as to how these findings on glutathione might be translated to a therapy? Might SLC25A39 or ATF4 be a target?
Dr. Birsov: We are actively trying to find small-molecule inhibitors, and we believe this may be helpful for certain cancers. Additionally, we need to know whether there will be off target effects as well.
MitoWorld: How did you become interested in mitochondria in the first place?
Dr. Birsov: We’re fascinated by how mitochondria connect metabolism to disease and cell function. These organelles contain thousands of metabolites, and yet, we still know very little about what most of them do or how their levels are maintained. Our work on mitochondrial transporters provides an entry point to start decoding this complexity, understanding how metabolites move across membranes and how these movements shape cellular behavior.
References
1 Yeh HW, DelGaudio NL, Uygur B, Millet A, Khan A, Unlu G, Xiao M, Timson RC, Li C, Ozcan K, Smith KW, Nascentes Melo LM, Allies G, Basturk O, Sickmann A, Byraktar EC, Possemato R, Tasdogan A, Birsov K (2025) Mitochondrial glutathione import enables breast cancer metastasis via integrated stress response signaling. Cancer Discovery 15: 2437–2449.
doi: 10.1158/2159-8290.CD-24-1556.
2 Wang Y, Yen FS, Zhu XG, et al. (2021) SLC25A39 is necessary for mitochondrial glutathione import in mammalian cells. Nature 599: 136–140.