Mitochondria Comes to the AACR: A Turning Point for Cancer Research and the Rise of Mitochondrial Transfer
AACR Annual Meeting 2026 — San Diego, California
“Mitochondrial Transfer Networks in Cancer Progression”
AACR Annual Meeting 2026 — San Diego, California
“What was striking was not simply that mitochondrial transfer was being discussed, but how many different cancer systems it appeared to connect. Across all three talks, mitochondria were presented less as isolated organelles and more as mobile biological assets shaping metastasis, immune exhaustion, therapeutic resistance, and even tumor innervation. You could feel the field beginning to connect previously separate observations into a larger systems-level framework.”
— Alex Sercel, Co-Founder, MitoWorld.org
For decades, mitochondria occupied an uneasy place in cancer research. Everyone knew they mattered. They appeared in discussions of metabolism, apoptosis, oxidative stress, and cellular energetics. But they rarely occupied center stage at major oncology meetings. The mitochondrion was often treated as background infrastructure — essential, certainly, but secondary to the “real” drivers of cancer biology.
Something changed at the American Association for Cancer Research (AACR) Annual Meeting 2026 in San Diego.
For the first time in the history of the AACR Annual Meeting, mitochondrial transfer in cancer was given its own dedicated symposium. That may sound procedural to outsiders, but to researchers working at the intersection of mitochondria, metabolism, immunology, and cancer, it represented something much larger: institutional recognition that mitochondrial dynamics are no longer peripheral to oncology. They are becoming central to it.
The symposium, “Mitochondrial Transfer Networks in Cancer Progression,” brought together three researchers approaching the field from distinct but converging directions:
- Yosuke Togashi, Okayama University
- Simon Grelet, University of South Alabama / Mitchell Cancer Institute
- Luca Gattinoni, University of Regensburg / Leibniz Institute for Immunotherapy (LIT)
Collectively, the presentations argued something profound: mitochondrial transfer is not a niche biological curiosity. It is emerging as a fundamental mode of intercellular communication in cancer biology — one that tumors exploit, immune systems respond to, and future therapies may intentionally manipulate.
After the session, MitoWorld.org reached out to all three panelists with a series of questions about the field, the symposium, and where mitochondrial cancer biology may be headed next. What emerged from those conversations was not just enthusiasm, but the unmistakable sense that an entire area of science is crystallizing in real time.
A Field Arrives
When asked what made the AACR symposium so important, all three researchers independently focused on the same point: this was a first.
“This was, to my knowledge, the first time mitochondrial transfer in cancer had been given its own dedicated special session at AACR,” explained Simon Grelet. “The room was charged with enthusiasm and curiosity.”
Yosuke Togashi emphasized the scale of interest from the oncology community itself:
“The sheer volume of questions indicated a significant surge in interest regarding how mitochondrial dynamics influence oncology.”
For Luca Gattinoni, the significance was not merely that the session existed, but how it was framed.
“The three talks together made an argument that no single one of us could have made alone: that mitochondrial transfer is not a niche phenomenon but a fundamental mode of intercellular communication that tumors exploit and that we can potentially exploit back.”
That framing matters.
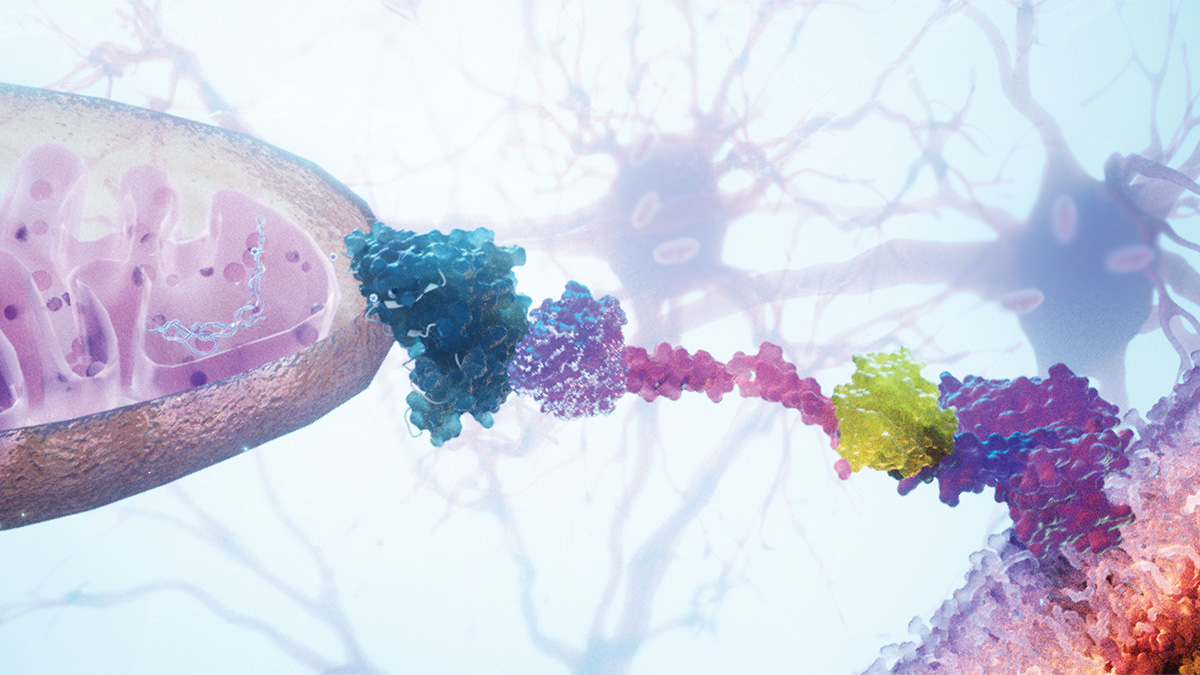
Cancer biology has increasingly become the biology of systems: tumor microenvironments, immune interactions, stromal signaling, metabolism, and cellular networking. What mitochondrial transfer research suggests is that mitochondria themselves may function as active biological currency moving between cells — influencing survival, adaptation, immune suppression, metastasis, and therapeutic resistance.
From “Power Plants” to Networks
One of the strongest themes emerging from the symposium was that the classical textbook view of mitochondria is rapidly collapsing.
For generations, mitochondria were taught primarily as intracellular energy factories — isolated organelles generating ATP inside sealed cellular boundaries.
That view now appears incomplete.
“The field is moving away from the traditional view of the mitochondrion as an isolated ‘power plant’ enclosed within a cell,” said Yosuke Togashi. “Instead, we are beginning to understand it as a dynamic component of a larger networking system.”
That conceptual shift may ultimately prove as important as any individual experiment.
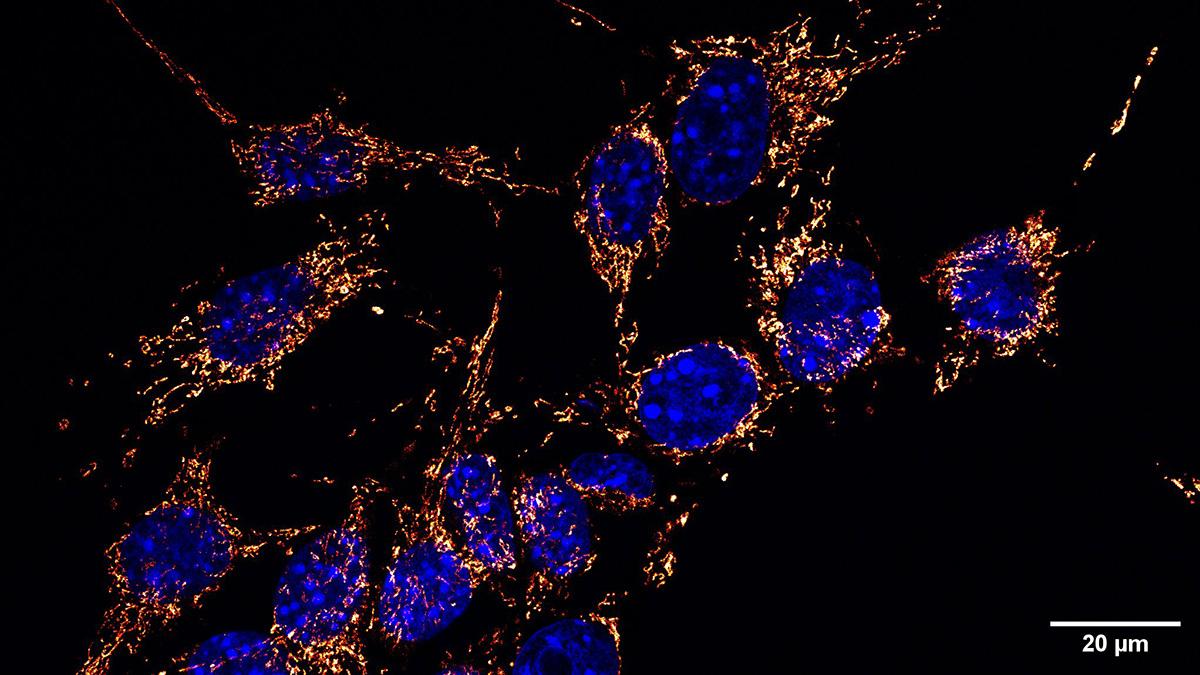
Mitochondria are increasingly being understood not simply as metabolic engines, but as signaling entities, stress sensors, inflammatory regulators, and now potentially mobile intercellular participants capable of moving between cells and altering biological outcomes.
Simon Grelet noted how rapidly evidence for mitochondrial transfer has accumulated across multiple dimensions of cancer biology:
“It is proving to touch multiple dimensions of cancer biology: how tumor cells acquire metabolic advantages, how they interact with their microenvironment, how they evade treatment, and more.”
Meanwhile, Luca Gattinoni highlighted how evidence is arriving simultaneously from very different domains:
“Nerve-tumor interactions shaping metastasis, tumor cells offloading dysfunctional mitochondria to suppress immune responses, stromal cells using the same mechanism to sustain T cell fitness.”
“The same biological currency,” he added, “deployed in radically different contexts.”
The Room Itself Told the Story
Scientific meetings often reveal the future of a field less through formal presentations than through hallway conversations afterward.
By all accounts, that happened here.
Yosuke Togashi was struck not only by the engagement during the session, but by what happened after it ended:
“The technical nature of the follow-up questions showed that researchers are already thinking about how to integrate these concepts into their own models.”
Luca Gattinoni described something rarer still:
“The conversation that emerged felt like the field thinking out loud in real time.”
That may ultimately be the most important signal of all.
Not simply that mitochondrial transfer research is growing, but that cancer researchers working in metastasis, immunotherapy, stromal biology, metabolism, and genomics are beginning to realize they are asking overlapping questions about the same underlying system.
The Next Frontier: mtDNA and the Mitonuclear System
If mitochondrial transfer itself is emerging as a major area, the next wave may center on mtDNA and the mitonuclear system.
The mitochondrial genome remains one of the least fully integrated components of modern cancer biology. Heteroplasmy, mutation dynamics, mitochondrial-nuclear coordination, and intercellular mitochondrial inheritance remain only partially understood.
Yosuke Togashi believes advancing sequencing technologies will rapidly change that.
“While the complexities of mutation patterns and heteroplasmy remain largely mysterious, advancing sequencing technologies will soon make this a focal point of cancer genomics.”
Luca Gattinoni sees the mitonuclear axis becoming its own frontier:
“We now have enough mechanistic footing to ask harder questions about directionality, selectivity, and what it truly means for a cell to absorb another cell’s mitochondria and their DNA.”
And perhaps most intriguingly, he believes the future will move beyond observation:
“The ambition should grow: not just observing transfer, but engineering it with intent.”
That single sentence hints at where this field could eventually lead: mitochondrial engineering, mitochondrial immunotherapy, and perhaps entirely new therapeutic architectures built around manipulating cellular energy and signaling systems directly.
A New Scientific Convergence
Historically, mitochondrial biologists and cancer researchers often existed in adjacent but separate scientific cultures. That separation may now be ending.
Simon Grelet noted that mitochondrial biology appeared across multiple AACR tracks far beyond this single symposium.
Meanwhile, Yosuke Togashi offered perhaps the most insightful observation of the entire discussion:
“The most vital ‘transfer’ occurring right now isn’t just between cells, but between disciplines.”
That may ultimately define this moment.
Cancer biology, immunology, mitochondrial medicine, metabolism, genomics, and systems biology are beginning to converge around a shared realization: mitochondria are not passive background organelles. They are dynamic participants in disease.
AACR 2026 may be remembered as one of the first moments when that convergence became visible at scale.
And if the energy in San Diego was any indication, this is only the beginning.