Confinement Causes a Mitochondria-Derived Nuclear ATP Surge
In a recent paper in Nature Communications, a multi-institute research team reported that cells confined by physical forces display several adaptations. They found that mitochondria accumulate at the nuclear periphery and produce additional ATP for use in the nucleus. These metabolic adaptations prepare the cell for DNA repair processes and cell proliferation.
Cells are occasionally subjected to physical forces that confine their shape and size. Cancer is a good example. Uncontrolled growth can result in physical confinement and pressures on organelles within the cell that can not only change the cytoskeleton but also the integrity of DNA and chromatin organization within the nucleus.
The research, led by Ritobrata Ghose and Fabio Pezzano, in the labs of Sara Sdelci and Verena Ruprecht, wanted to better understand how the physical tissue microenvironment affects cells and the inherent adaptation mechanisms in place to buffer against microenvironmental effects. In particular, they focused on the effects on the nucleus and its proximate organelles. Using an unbiased approach, they discovered a unique functional link between the nucleus and the mitochondria. After the discovery, the team went on to explore this relationship in live cells by high-resolution microscopy.
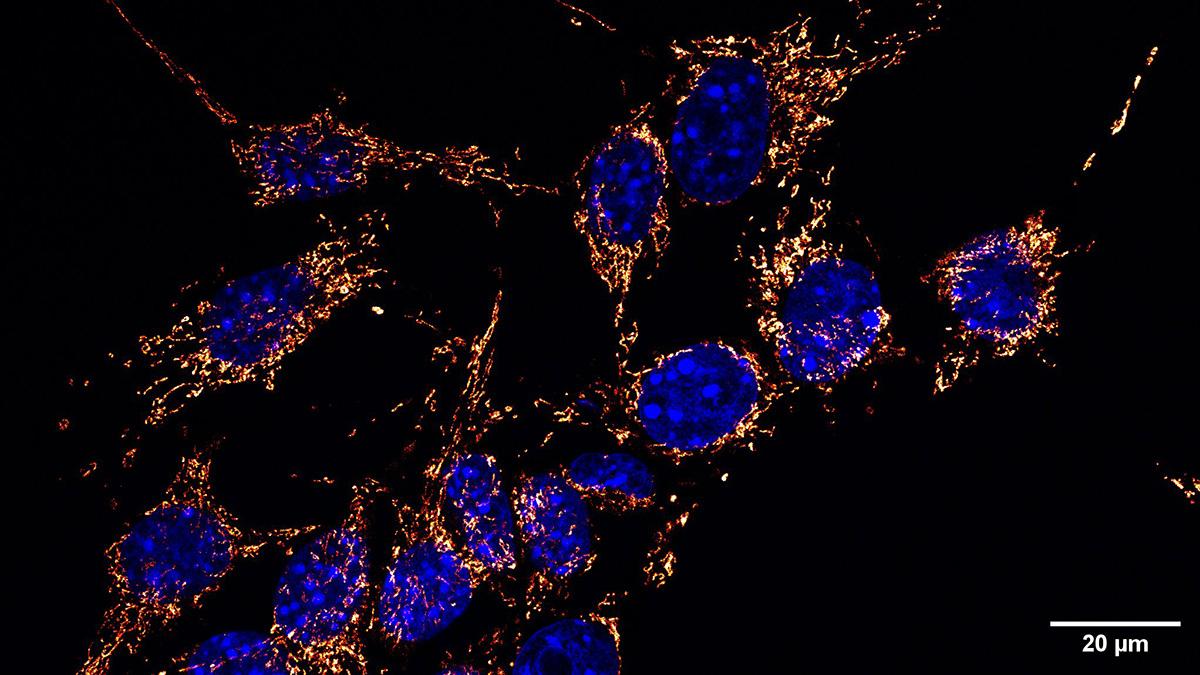
Interestingly, they found that sustained physical confinement caused mitochondria to reposition themselves at the nuclear membrane and within regions of nuclear deformation. This repositioning was facilitated by the actin cytoskeleton and an endoplasmic reticulum-derived net that pulls them closer to the nuclei. Functionally, this nucleus-mitochondria association or proximity yielded a surge in the amount of ATP available within the nucleus, which is critical for genomic material integrity.
This paper describes a complex interaction of multiple factors that result from the physical confinement of the organelles within a cell. Cells respond by gathering the mitochondria close to the nuclear membrane so that an ATP surge can prepare the cell for DNA repair and cell proliferation after the stress is relieved.
Statement of Significance: Ritobrata Ghose, Ph.D.
This discovery emerged from an organic and energizing interdisciplinary collaboration between the Sdelci lab’s expertise in nuclear metabolism and the Ruprecht lab’s in biophysics. We believe interdisciplinary sciences will underpin the next wave of major discoveries, and we’re proud to have been at the forefront of it.
A Discussion with Ritobrata Ghose:
MitoWorld: What direction might your research take to further expand this work?
Our current work has demonstrated a mechano-metabolic axis which regulates chromatin state, chromatin integrity and cell fitness. This intersection between nuclear biophysics, nuclear metabolism, and epigenetic and transcriptional programs offers a unique and exciting opportunity that promises to reveal previously unimagined regulatory links. Further exploration of this intersection, in both disease and normal physiological conditions, will expand our understanding of fundamental cell biology.
MitoWorld: Can you speculate on the signaling molecules and receptors in the mitochondria and nucleus that might be involved in controlling the actions noted in your paper?
Dr. Ghose: This is one of the most exciting avenues for future exploration. We have identified several factors involved in NAM formation, including the relevance of mitochondrial shape and fusion dynamics. Whether mitochondria fuse with structures beyond themselves remains an intriguing open question. We also observe that the ATP surge is exclusive to the nucleus, suggesting directional shuttling of ATP, possibly through dedicated channels, which warrants further investigation.
MitoWorld: Actin and microtubules are important player in this system. How do you think they are controlled?
Dr. Ghose: Actin plays a critical role in regulating the cellular response to confinement. As cancer cells become migratory and aggressive, they are known to exhibit increased actomyosin contractility, for instance through ROCK-myosin II signaling, which enhances their ability to respond to complex mechanical cues. Consistently, we observed that more aggressive cells, particularly those at the invasive front, displayed significantly higher NAM formation.
Interestingly, the role of microtubules appears to be more nuanced. While microtubules are well-established drivers of mitochondrial transport, we found that depolymerization of microtubules using Nocodazole actually enhanced NAM formation under confinement. This suggests that rather than promoting mitochondrial accumulation at the nucleus, microtubules may normally act to restrict it. Understanding how the balance between actin and microtubule-based mitochondrial positioning is regulated will be an important next step.
MitoWorld: This interesting study has implications for the evolution and development of multicellular organisms. Physical confinement might be one factor that helps them maintain their place and size, but that cancer leads them to forget. Do you have any thoughts on this?
Dr. Ghose: It is likely that these mechano-metabolic adaptations are a conserved cellular response to physical challenge. In our work, we observed this mechanism across multiple cancer types, suggesting it operates across diverse tissue contexts. This points to a fundamental and evolutionarily conserved adaptation, with cancer representing a corruption of these normal cellular programs rather than the emergence of entirely new ones. Immune cells, for instance, are highly motile, constantly traversing physically demanding environments, and would be an interesting model to explore these mechanisms in. Understanding how confinement-induced ATP dynamics operate in such cells, and how they differ in the disease context, could open exciting new directions beyond cancer biology, but also help understand therapeutic vulnerabilities.
MitoWorld: Might your findings have any clinical relevance? Can you envision ways it might be harnessed for future treatment strategies?
Dr. Ghose: We validated our discovery of this mechano-metabolic adaptation in patient tumors and particularly at the invasive front of tumors, revealing a clear dependency and therefore a potential vulnerability of aggressive and metastatic cancer cells. As we deepen our mechanistic understanding of what features are particularly enriched within cancer cells, we expect to identify actionable targets and evaluate their therapeutic potential.
Reference
Ghose R, Pezzano F, Badia R, Kourtis S, Sheraj I, Das S, Gañez Zapater A, Ghose U, Musa-Afaneh S, Espinar L, Coll-Manzano A, Parapatics K, Ivanova S, Sànchez-Fernàndez-de-Landa P, Radivojevikj D, Venturini V, Wieser S, Zorzano A, Müller AC, Ruprecht V, Sdelci S (2025) Mitochondria-derived nuclear ATP surge protects against confinement-induced proliferation defects. Nat Commun 16(1): 6613.
doi: 10.1038/s41467-025-61787-x.