Mitochondria and Sleep
All of us need sleep, but the reasons for that need are poorly understood. Scientists have previously reported changes that occur in the brains of animals that have remained awake for long periods. However, it wasn’t clear if these changes are causes or results, and the physical basis of the need for sleep is still unknown.
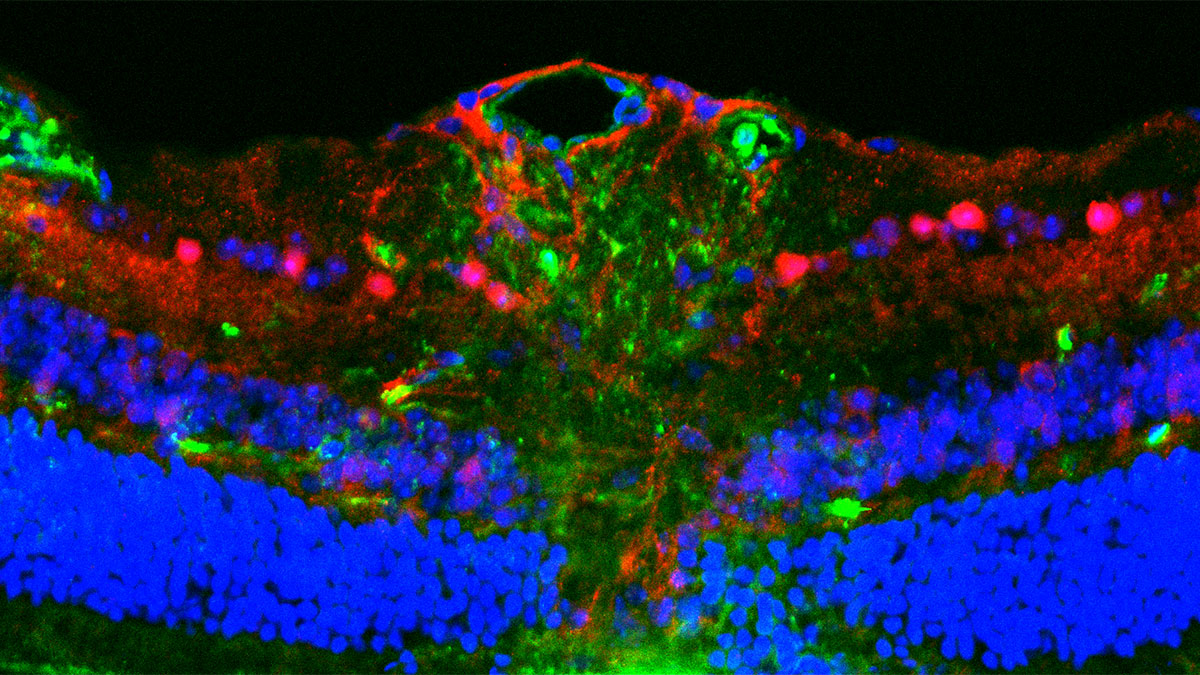
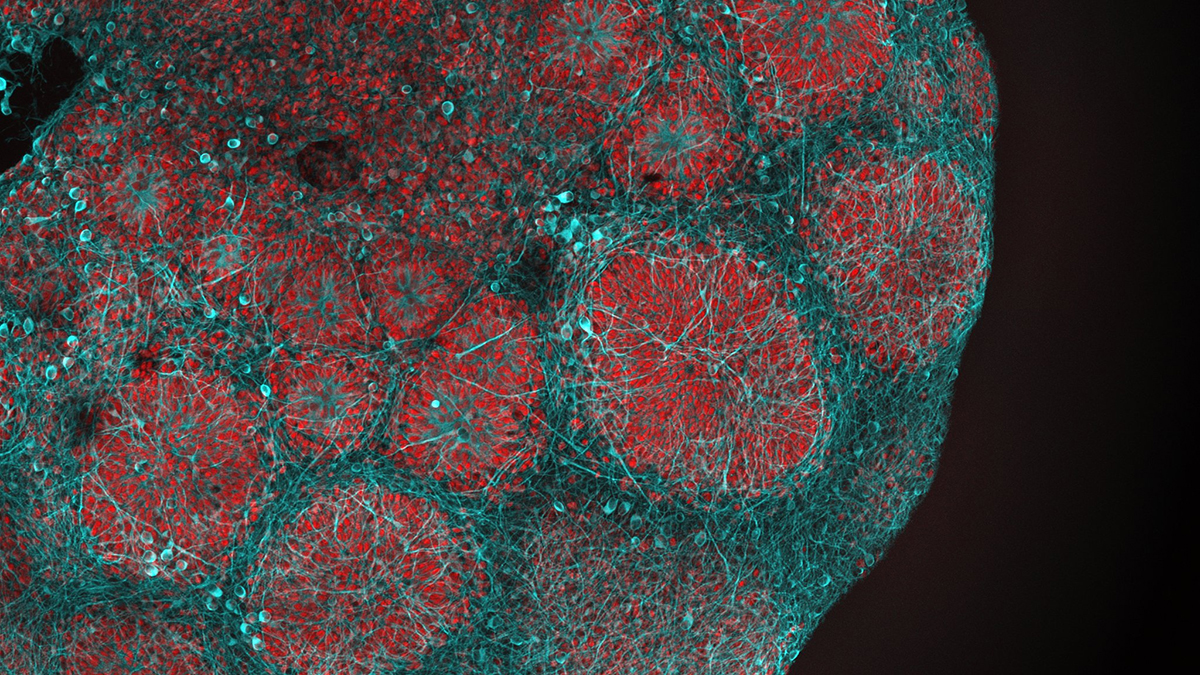
In a recent paper published in Nature, researchers at Oxford University, led by Gero Miesenböck, argued that the only way to sort out this issue was to look at specialist neurons with known roles in inducing and maintaining sleep. To gain insights into what makes us sleepy, the Oxford scientists studied the fruit fly Drosophila and compared the genes turned on or off in sleep-promoting neurons of rested and tired flies. Interestingly, almost all the upregulated genes encoded proteins involved in mitochondrial respiration and ATP synthesis. Other effects of sleep loss included mitochondrial fragmentation, mitophagy, and more contacts between mitochondria and the endoplasmic reticulum. After sleep, all of these were reversed, and nearly everything returned to normal.
The Miesenböck team speculate that the reason why animals need sleep is their power-hungry nervous systems, whose energy demands can only be met by stripping electrons from foodstuffs and transferring them to oxygen. Oxygen, however, is a double-edged molecule because its chemical nature invites missteps in mitochondrial electron trades; electrons leak from the respiratory chain and form damaging reactive oxygen species. This, they suggest, is the root cause of sleep.
The authors also note that neurons regulating sleep and energy balance work through similar mechanisms. These mechanisms involve cycles of mitochondrial fission and fusion. Fusion increases weight gain and fat deposition and also sleep. Hindering fusion does the opposite.
They conclude that sleep, like aging, may be an inescapable consequence of aerobic metabolism. Their findings enhance the understanding of how sleep is controlled and why we need sleep. The deeper understanding of sleep might also eventually point to potential new strategies for helping patients with sleep challenges.
A Conversation with Dr. Miesenböck
MitoWorld: What do you see as the next steps in your research?
There are two obvious follow-up steps. The first is to ask whether mitochondrial respiration also regulates mammalian sleep, or whether this is a fly thing. I definitely think it is not, but it still needs to be proven formally. The second question relates to the function of sleep. We may have put our finger on the cause—electrons spilling from mitochondria—but how sleep prevents or repairs the resulting damage remains unexplained.
MitoWorld: Multiple theories try to explain the need for sleep. Your work adds real observations to the idea of sleep as an ancient metabolic need for energy-consuming brains. Is this likely to be enough to tip the scales in favor of mitochondria?
Maybe there are no scales to be tipped because sleep has many functions and most theories will turn out to be correct. In our view, sleep originally evolved because neurons consume a lot of oxygen to support their energy needs. Once mitochondria had forced rest periods on animals, other functions were likely added, such as the downscaling of synapses strengthened during waking or memory consolidation.
MitoWorld: You described how you deprived the flies of sleep in your methods section, but I can imagine that readers might be wondering how you keep flies from sleeping. Could you briefly summarize that process and also how you induced sleep in the flies?
We deprived the flies of sleep just like we would deprive you: by poking them every 10 seconds or so. Fortunately, we have a machine that does that.
MitoWorld: You note a connection between sleep and eating and the need to maintain a balance for each of these key physiological processes. Can you elaborate on that?
Both sleeping and eating are, to use an old-fashioned term, drives: basic needs regulated by feedback. The less you sleep, the more tired you are; the less you eat, the more hungry you are. We now know that the neuronal controllers of sleep and hunger are similar in that they both depend on cycles of mitochondrial fission and fusion.
MitoWorld: This study features some very nice technical work (e.g., selection of an organism with cells known to be involved in sleep, a key fluorescent marker for selection, single-cell transcriptomes). Can you comment on those?
Thank you. My favorite of the many clever experiments Raffaele Sarnataro did draws on the concept of optogenetics, which we originated a quarter of a century ago. Raffaele put a light-driven proton pump into the mitochondria of sleep-promoting neurons and powered ATP synthesis with photons rather than electrons. This made electrons redundant and put flies to sleep.
MitoWorld: Could you speculate on whether your findings can be extrapolated to humans?
There are some hints. For example, an overwhelming sense of tiredness is a common symptom of human mitochondrial disease, despite normal ATP levels and no muscle fatigue.
MitoWorld: How did you come to be interested in mitochondria in the first place?
A previous study from my lab (Kempf et al., Nature 2019) provided the first clue that mitochondria are involved in the regulation of sleep. We discovered that sleep loss increases reactive oxygen species (ROS) in sleep-control neurons and that curbing electron leakage in their mitochondria (which fuels ROS production) reduces the pressure to sleep. In the present study, we played dumb and pretended we knew nothing about how sleep-control neurons sense the need for sleep. And voilà, when we looked at gene expression changes with a completely open mind, mitochondria popped up again.
Reference
Sarnataro R, Velasco CD, Monaco N, et al. (2025) Mitochondrial origins of the pressure to sleep. Nature 645: 722–728. https://doi.org/10.1038/s41586-025-09261-y