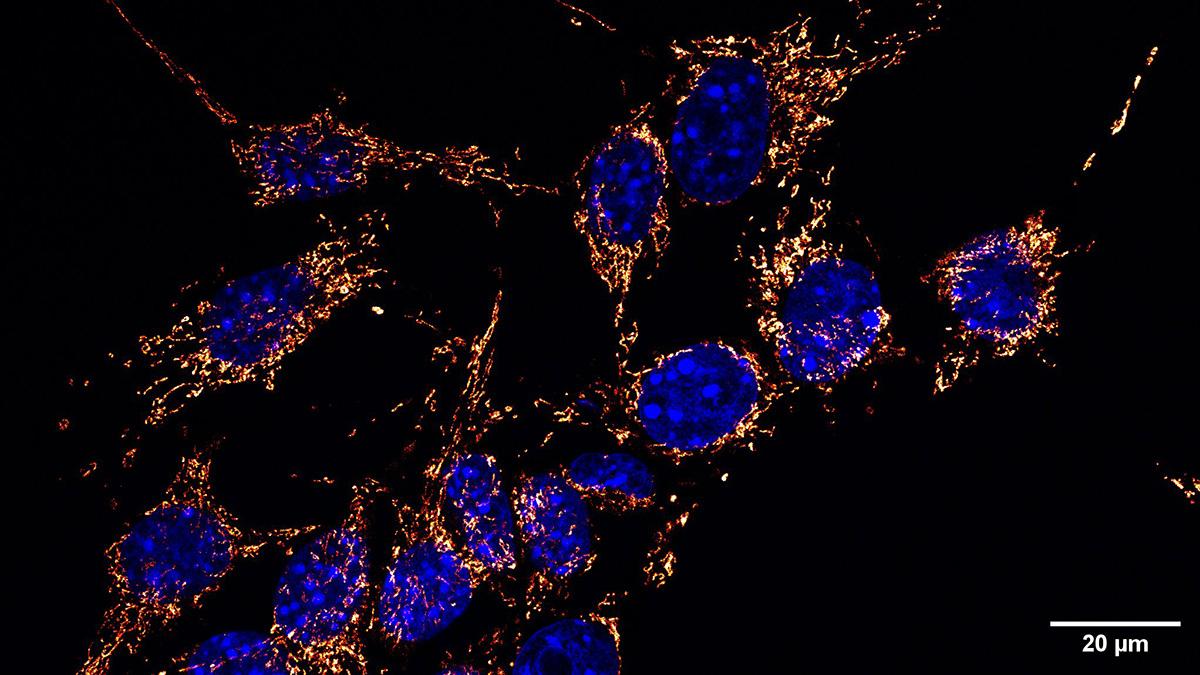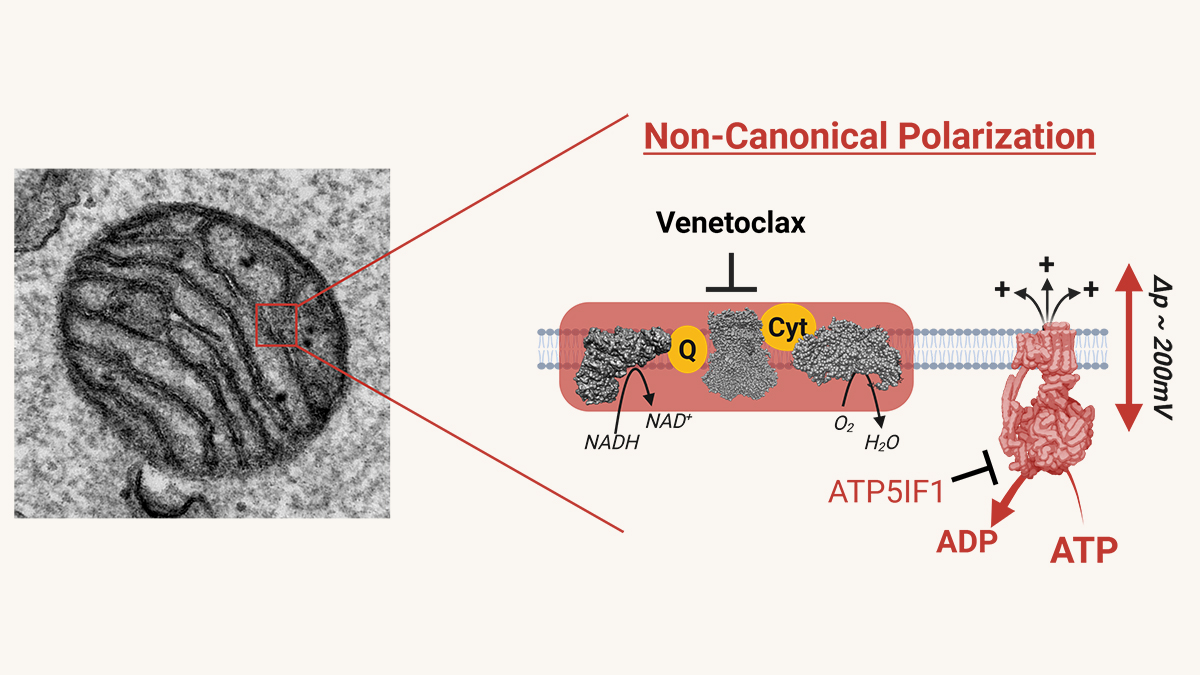
Left: TEM image of a single mitochondrion in an MV4-11 AML cell. Right: AML cells maintain mitochondrial membrane potential via ATP hydrolysis by reversing ATP synthase, a dependency enhanced by venetoclax and targetable through ATPIF1 expression.
Potential ATP-Centered Treatment in Acute Myeloid Leukemia
A multi-institute research team led by Kelsey H. Fisher-Wellman describes a unique mechanism in mitochondria that might be a therapeutic target in patients with acute myeloid leukemia (AML). The paper was published in Science Advances.
Cancers are thought to need large amounts of energy, and thus, oxidative phosphorylation is a tempting target for cancer therapies. Unfortunately, the attempts to find therapies that could differentiate between healthy and diseased mitochondria have met with limited success. For example, the FDA approved cancer therapy, venetoclax, which inhibits BCL-2 and mediates BAX/BAK-dependent apoptosis, has limited effectiveness due to the development of chemoresistance. Mitochondrial polarization of resistant AML cells is unaffected.
The group led by Dr. Fisher-Wellman noted that AML mitochondria have an action that differs from healthy blood cells. Healthy cells connect ATP synthesis to respiration. However, AML cells maintain inner membrane polarization by consuming ATP. The researchers wondered if the AML cells used this reversal of the ATP synthase reaction to resist chemotherapy. They set out to test this possibility. They discovered a central role for ATP51F1, an F1-ATPase inhibitor. Overexpression of this enzyme heightened sensitivity to venetoclax, and its knockdown venetoclax resistance.
These findings further our understanding of the bioenergetics of AML cells and the relationship between oxidative phosphorylation and AML. This work is especially important since AML relapses have such a dire prognosis. Importantly, they implicate matrix ATP consumption as a potential cancer cell-specific target for inhibiting the development of chemoresistance in AML.
Statement of Significance from Dr. Fisher-Wellman
In AML cell lines and primary patient samples, we discovered that leukemic mitochondria are deficient in their ability to sustain mitochondrial membrane potential through respiratory flux. Maintenance of membrane potential is essential for cell survival. To compensate, AML cells engage a non-canonical—and possibly evolutionarily ancient—bioenergetic strategy: they reverse the ATP synthase enzyme. Rather than using membrane potential to drive ATP production, AML cells hydrolyze ATP to sustain mitochondrial polarization. This distinction is profound. Normal cells do not typically rely on ATP synthase reversal to maintain membrane potential. As a result, this AML-specific mitochondrial wiring represents a near-ideal cancer-selective vulnerability. Importantly, this pathway is most active in drug-resistant AML populations—the very cells responsible for relapse and treatment failure.
Conversation with Dr. Fisher-Wellman:
MitoWorld: Can you give us some idea of how you intend to further advance the results of the current work?
Fisher-Wellman: Most cancer therapies are built around exploiting a dependency or vulnerability that is unique to malignant cells. Our work demonstrates that AML cells maintain mitochondrial membrane potential through a mechanism that is distinct from most normal tissues. We believe this cancer-specific difference represents a therapeutically actionable vulnerability.
MitoWorld: It is interesting that the AML mitochondria have this particular weakness. Do you have any speculation on why it is? Is it possible that other cancers might share this?
Fisher-Wellman: Sustaining mitochondrial membrane potential through ATP hydrolysis is generally viewed as a hallmark of bioenergetic ‘sickness’, typically observed in stressed or damaged cells as a survival mechanism. Our data, together with emerging evidence from the field, indicate that AML mitochondria exist in a persistently ‘sick’ state. Importantly, this bioenergetic liability represents a vulnerability that may be selectively exploited for therapeutic benefit.
MitoWorld: Do you have any plans to pursue the development of the potential clinical relevance of your discovery?
Fisher-Wellman: We are actively translating this discovery into mitochondria-targeted therapeutic strategies that exploit the distinct mechanisms by which AML cells maintain mitochondrial membrane potential.
MitoWorld: Mitochondria seem to be implicated in many diseases. Part of the reason is clearly its fundamental activity in energy production. However, they have assumed key roles in many other cellular functions. As a mitochondria researcher, are you as fascinated by these observations as we are?
Fisher-Wellman: That is an unequivocal yes. My entire research career has been focused on understanding how alterations in this organelle cause and contribute to some of humanity’s most formidable diseases.
MitoWorld: How did you come to be interested in mitochondria? Was it the energy requirements of cancers that connected you to the energy source?
Fisher-Wellman: I have studied mitochondria throughout my entire career and developed a deep appreciation for the remarkable heterogeneity that exists within this organelle across the body’s cells. It was this diversity that ultimately drew me to cancer, where I recognized that vast unexplored biology remained to be discovered.
Reference
Hagen JT, Montgomery MM, Aruleba RT, Chrest BR, Krassovskaia P, Green TD, Pacheco EA, Kassai M, Zeczycki TN, Schmidt CA, Bhowmick D, Tan S-F, Feith DJ, Chalfant CE, Loughran Jr. TP, Liles D, Minden MD, Schimmer AD, Shakil S, McBride MJ, Cabot MC, McClung JM, Fisher-Wellman KH (2025) Acute myeloid leukemia mitochondria hydrolyze ATP to support oxidative metabolism and resist chemotherapy. Science Advances 11(15): eadu5511.
https://www.science.org/doi/full/10.1126/sciadv.adu5511