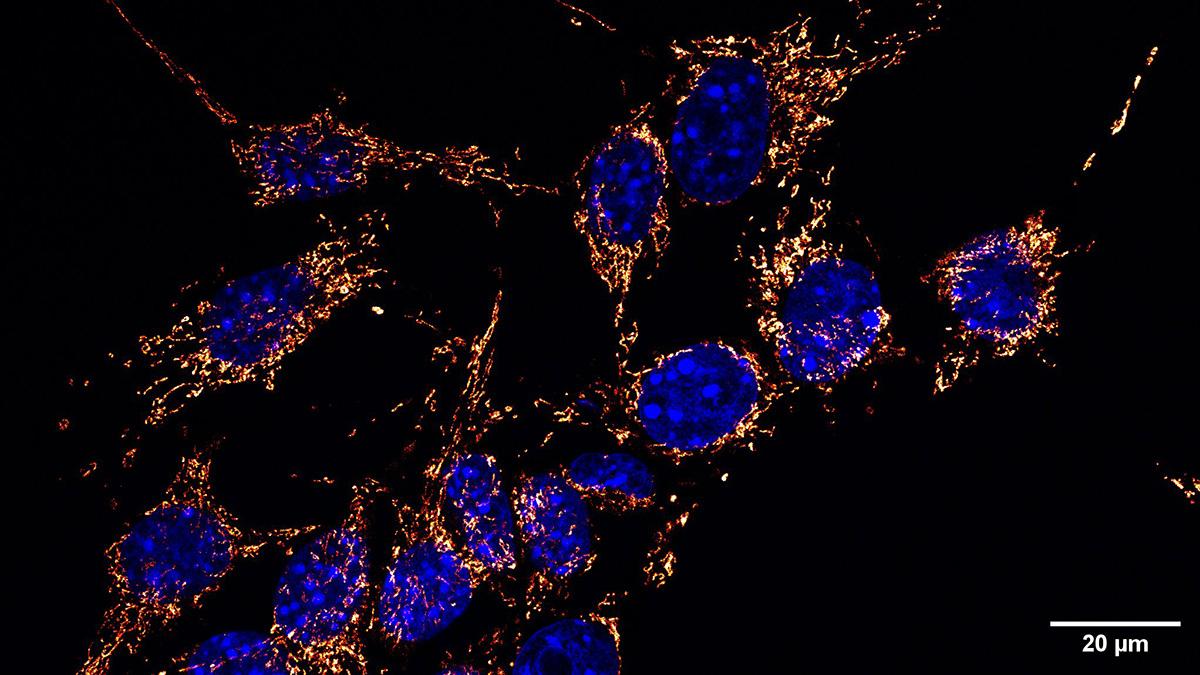
Karolinska Institutet, Solna Campus, Photographer: Ulf Sirborn
Quality Control of Mitochondrial DNA Transmission
Unlike the nuclear genome, which contains genes from both parents, the mitochondrial genome is inherited only from the mother. This presents a problem since mitochondrial DNA (mtDNA) is prone to mutations that can accumulate over generations and threaten species survival. To avoid this, two still poorly understood mechanisms have been selected for in evolution to counteract the accumulation of harmful mtDNA mutations: the bottleneck, whereby only a small number of mtDNA molecules is passed on to the next generation and purifying selection, a mechanism that prevents mtDNA molecules with severe mutations from being transmitted.
A recent study published in Science Advances sought to better understand how these two mechanisms cooperate to ensure healthy mitochondria in offspring. The research was led by Professor Nils-Göran Larsson, MD, PhD, and performed at the Department of Medical Biochemistry and Biophysics at Karolinska Institutet, Sweden. Dr Laura Kremer, first author of the study is currently an independent researcher at University of Göttingen and was conducting the research as a postdoc at Karolinska Institutet.
To explore the role of the bottleneck and purifying selection in the transmission of mutant mtDNA, the team produced mouse models with random mtDNA mutations along with alleles that modulated mtDNA levels or decreased autophagy.
They found that tightening the bottleneck, by decreasing the amount of mtDNA in the mother, resulted in greater variability in the levels of mtDNA mutations between individuals but reduced the overall mutational burden. Vice versa, decreasing autophagy weakened the selection, allowing more mutant mtDNA molecules to be passed on to the offspring leading to an increased mutation burden.
This study yielded novel insight by showing that the two mechanisms directly interact. Learning how cells protect the mitochondrial genome is valuable not only from an evolutionary perspective but also has medical relevance. Mutations in mtDNA are associated with many human diseases, including mitochondrial disorders, cancer, neurodegeneration, and ageing.
A conversation with Dr. Larsson
MitoWorld: You have a prolific laboratory, and this paper represents a great deal of work. Perhaps the work in this paper has progressed since its publication. Could you summarize the direction of any newer work?
Larsson: Since the publication, one direction we have undertaken involves the development of new mouse models that allows us to track different type of mtDNA variants and understand whether there are differences in the selective mechanisms that limit their transmission.
MitoWorld: At first glance, a reader might assume that increasing heteroplasmy would lead to more mtDNA mutations being transmitted. Can you simplify that concept?
Larsson: It may seem intuitive that higher levels of mutant mtDNA would facilitate mutation transmission, but our study shows that purifying selection is more efficient if mutation levels increase. A tighter bottleneck increases the heteroplasmy variance between individuals, but it also allows selection to act more efficiently. In other words, harmful variants can be identified and purged more effectively when individual mutations are present at higher levels.
MitoWorld: Can you expand on what triggers autophagy? It’s interesting that the one protein Bcl2l13 attaches mitochondria with a high HF ratio to the autophagosome.
Larsson: Autophagy is triggered by several cellular stress signals, nutrient limitation, energy imbalance, or structural damage to the organelles. In the germline, we think the process is tuned specifically to identify dysfunctional mitochondria. Bcl2l13 appears to act as a recognition factor, marking mitochondria with a high proportion of mutant genomes for degradation. This selective role is still only partly understood, but it suggests a remarkable level of specificity during mtDNA transmission.
MitoWorld: The conservation of aspects of these mechanisms across most metazoa is interesting. Most eukaryotes feature maternal inheritance of mitochondria. These mechanisms must have evolved very early in evolution. Any speculation?
Larsson: Yes, these systems likely arose very early. Once organisms started relying on oxidative phosphorylation, mutations in the mitochondrial genome became a genuine threat to fitness. Mechanisms such as bottlenecks and selective degradation would have been strongly favored by evolution. Their conservation suggests they were effective solutions and have remained largely unchanged across hundreds of millions of years.
MitoWorld: Your extensive work on mitochondria has covered many topics, including cancer, inflammation, obesity, and much more. Is the central role of mitochondria in all these areas a function of energy production or are there other activities involved?
Larsson: Energy production is certainly fundamental, but mitochondria do far more. They influence signaling pathways, metabolite synthesis, innate immunity, and cell death. Their dysfunction sends ripple effects through many biological systems, so it is not surprising that mitochondrial defects appear across diverse diseases. Understanding mtDNA inheritance is one aspect of a much broader picture of mitochondrial biology.
MitoWorld: Can you imagine how your findings might be translated into clinical treatments? Will your lab be following up on that possibility?
Larsson: We are cautious about translating findings prematurely, but the work does suggest conceptual avenues. If we can identify the key factors that guide purifying selection, it may eventually be possible to enhance this process in patients carrying pathogenic mtDNA variants. Our immediate focus remains on basic mechanisms, but we collaborate closely with clinical groups, and we certainly see therapeutic potential in the long term.
MitoWorld: In you brief bio on the MitoWorld website, you stated that you became interested in mitochondrial DNA in diseases during your PhD training. Since then, you have contributed significantly to the understanding of mitochondria. Could you comment on your thoughts on these organelles today?
Larsson: My appreciation for mitochondria has only deepened over the years. They are extraordinarily dynamic organelles, genetically, metabolically, and evolutionarily. What continues to inspire me is how much remains unknown. Every time we think we understand a pathway, we find additional complexity. That curiosity is what keeps our field vibrant, and I believe mitochondria will continue to surprise us for many years to come.
Reference
Kremer LS, Golder Z, Barton-Owen T, Papadea P, Koolmeister C, Chinnery PF, Larsson NG (2025) The bottleneck for maternal transmission of mtDNA is linked to purifying selection by autophagy. Science Advances 11(46): eaea4660.