Cell-Type-Targeted Mitochondrial Transplantation
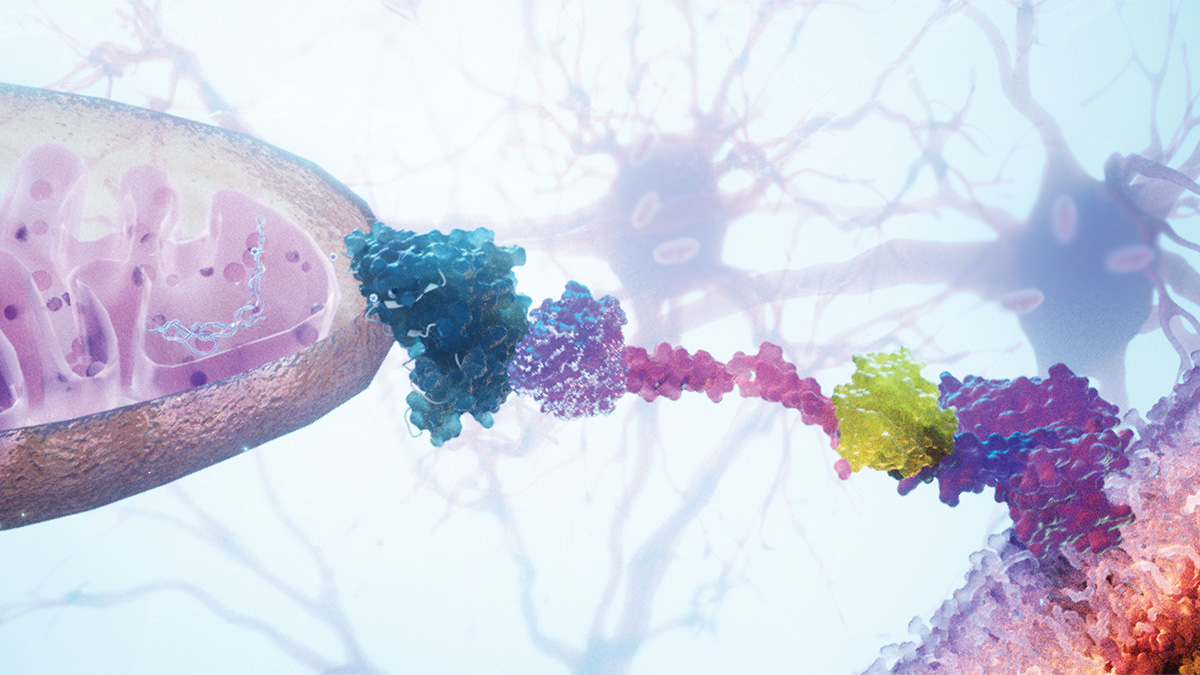
Dysfunctional mitochondria are associated with serious diseases (e.g., neurodegenerative disorders, heart failure), and transplantation of healthy mitochondria to diseased cells has been suggested as a possible therapeutic strategy. Recently, a research team, led by Botond Roska, developed a system for transplanting mitochondria to specific cell types. Their MitoCatch system is an exciting advance in harnessing mitochondria in therapies. The study was published recently in a paper in Nature.
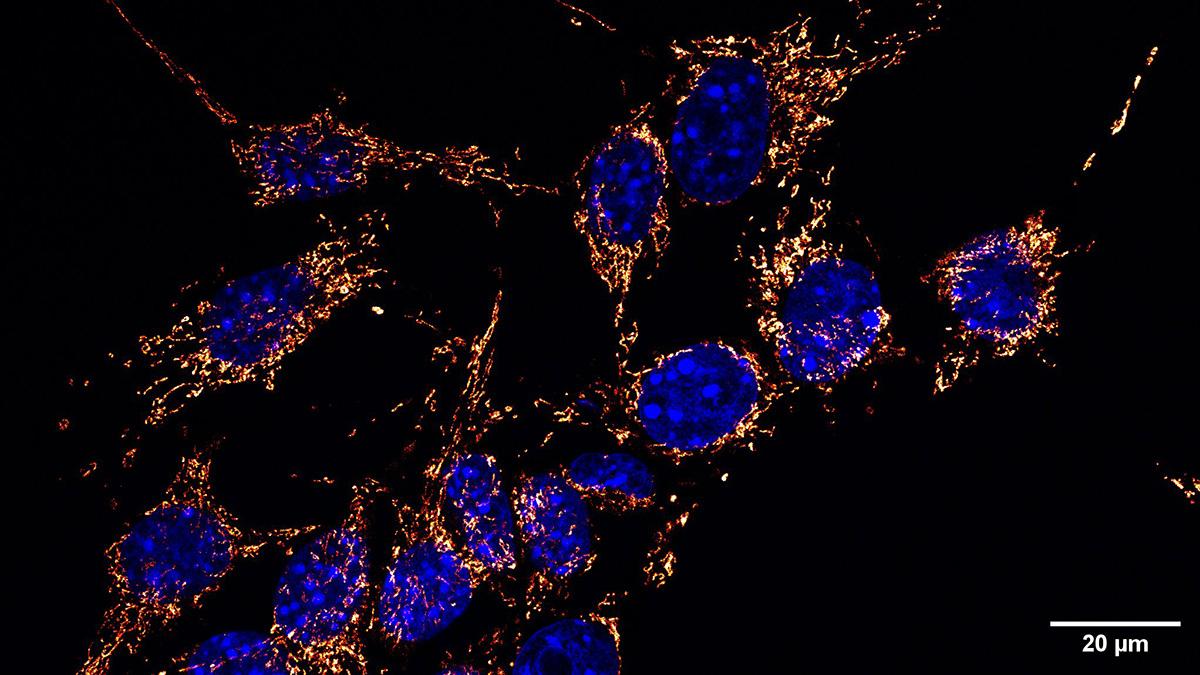
The Roska team sought to identify protein binders that would facilitate the uptake of donor mitochondria by specific target cells. They engineered a series of such binders for their MitoCatch system. Using this system, they showed that the donor mitochondria were internalized into the cytosol and that they behaved as normal mitochondria exhibiting movement, fusion, and fission. In addition, the researchers showed that the mitochondria could be targeted to specific cell types, including retinal, cardiac, endothelial, and immune cells, and neurons. Most importantly, the diseased or damaged cells receiving the donor mitochondria had improved survival and function.
In a relatively short time, mitochondrial transfer has moved from fantasy to reality. The development of MitoCatch now provides a method for targeting specific disease cell types. This exciting advancement brings mitochondrial transfer closer to a therapeutic strategy for serious disease conditions.
A Statement of Significance from Dr. Roska:
Many diseases that involve malfunctioning mitochondria currently have no effective treatment. Transplanting healthy mitochondria into diseased cells has emerged as a promising therapeutic approach, but until now, there has been no reliable way to deliver them to the right cell types in the body. We developed a system called ‘MitoCatch’ that uses protein-based targeting tools to guide healthy mitochondria directly to affected cells. In laboratory experiments with human cells and in live mice, MitoCatch-delivered mitochondria improved the survival of damaged nerve cells. MitoCatch thus offers a new strategy for treating diseases linked to mitochondrial dysfunction by delivering healthy mitochondria precisely where they are needed.
A Conversation with Dr. Roska:
MitoWorld. Your work offers lots of intriguing possibilities. Can you give us an idea of where you are going next to follow up on this paper?
Dr. Roska: We are particularly interested in understanding how long donor mitochondrial DNA can persist in recipient human cells, as this information is important for therapy development. We are also optimizing our bispecific protein binders for specific human applications.
MitoWorld. Your work also has obvious clinical possibilities. Do you plan to exploit those as well?
Dr. Roska: Yes. Our goal is to develop MitoCatch into a therapy. We are in discussions with leading physicians across different medical fields to identify which mitochondrial diseases would be the best fit for the first MitoCatch-based treatments. LHON is among the first candidates.
MitoWorld. We have been following the emergence of mitochondrial transfer for some time. Are you as amazed as we are about how quickly it has been accepted as a fact and as a potential therapy?
Dr. Roska: This is indeed a fascinating field that is moving ahead at lightning speed. There are still sceptics who question the usefulness of mitochondrial transfer as a therapy, but this is healthy in science. The best way to respond to skepticism is to demonstrate the value of mitochondrial transfer in the clinic.
MitoWorld. In the Discussion, you mention that your system, like mitochondria free in the blood, did not elicit an immune response upon transfusions. That’s an interesting observation for your work and evolution. Can you speculate on that?
Dr. Roska: The key question is what these free mitochondria in the blood are doing — whether they are functional or waste products. This is not yet known, and the answer will have important implications for therapy.
MitoWorld. Clearly, mitochondria are the focus of MitoWorld, and we also enjoy hearing what brought researchers to the study of these amazing organelles. Can you tell us how you became interested in mitochondria?
Dr. Roska: About eight years ago I was thinking about the complexities of different therapeutic modalities. Gene therapy introduces one or at most a few genes into the body and so carries very low genetic complexity. Cell therapy, on the other hand, brings all of a cell’s genes into the body and, therefore, operates at extremely high complexity. I wondered whether therapies with an intermediate level of genetic complexity were possible. This led me to think about organelles and mitochondria in particular. At the time, very few papers had been published on mitochondrial transfer, but this has since changed dramatically, and I am very happy to be part of such an exciting and fast-growing field.
Reference
Ayupov T, Moreno-Juan V, Curtoni S, Fratzl A, et al. (2026) Cell-type-targeted mitochondrial transplantation rescues cell degeneration. Nature 15: