Mitochondrial, Inflammatory and Neuronal Dysfunction in Bipolar Disorder
In a recent study by Dr. Ana Andreazza of The University of Toronto, a team of researchers generated 3D brain organoids from patient blood cells to study how cells from patients with bipolar disorder differed from those without. Culturing these brain organoids from 3 normal controls and 3 patients with bipolar disorder (BD), they find significant metabolic and immune differences that impact neuronal function. The study published in Translational Psychiatry points to mitochondria as key players in this complex psychiatric condition.
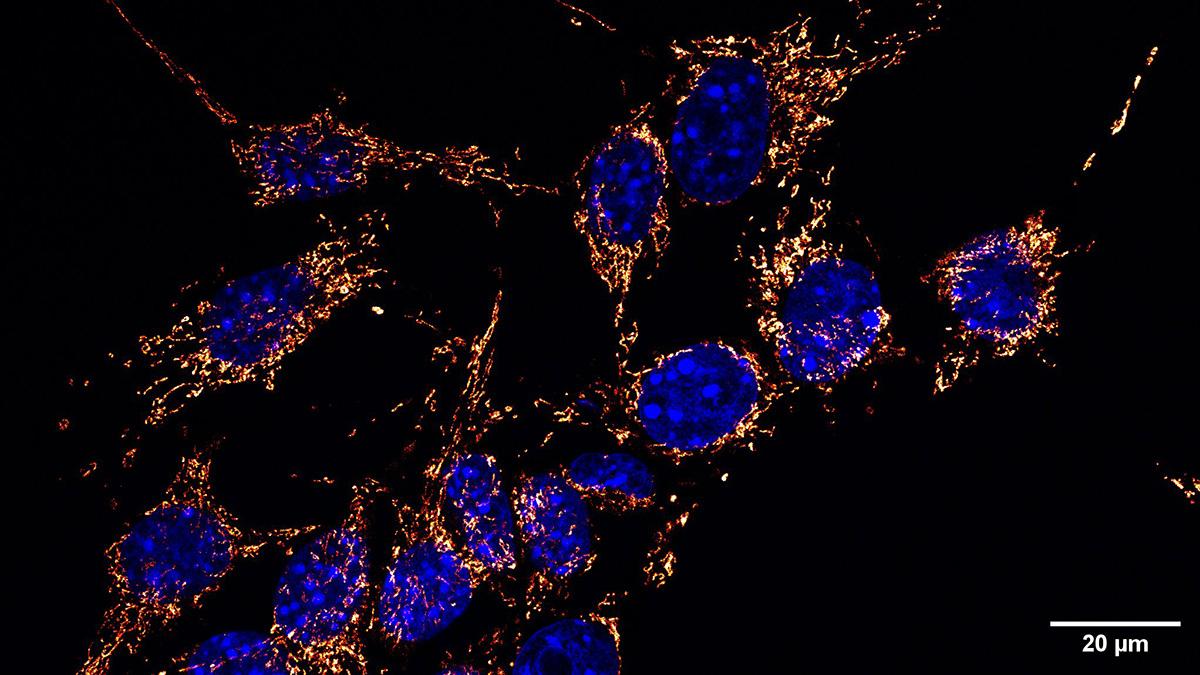
Comparing BD-derived brain organoids to normal controls, Andreazza finds many abnormalities. While their structure, organization and cell types are normal (consisting of neurons and astrocytes), their metabolic function is impaired. Deficits in ATP within these cells can be traced back to dysfunctional mitochondria, which exhibit altered morphology and weaker membrane potentials. BD brain organoids have small, rounded mitochondria, indicative of immature organelles experiencing oxidative stress and fragmentation. Indeed, these mitochondria show deficits in membrane potential and polarization, which are essential to generate ATP. As a result, these cells are not able to keep up with the high energy demands of neurons in the brain, leading to misfiring and hyperactive neural networks that are a hallmark of BD. This energy deficit also impairs neurogenesis, with BD brain organoids containing fewer cells than controls.
Not only do dysfunctional mitochondria impact brain function via energy deficits, but they also drive a neuroinflammatory cascade that further damages neurons. When mitochondria are stressed they release reactive oxygen species (ROS) and mitochondrial DNA (mtDNA) into the cell, both of which are elevated in the BD brain organoids compared to controls. These molecules trigger innate inflammatory pathways, including inflammasome activation, that drive neuroinflammation and pathology in a self-reinforcing cycle. These impacts are observed in neurons and astrocytes, supporting cells that promote neuronal health but can perpetuate inflammatory damage when dysregulated.
Many of Andreazza’s findings in brain organoids parallel observations in patients with BD. For example, high ROS and mtDNA are associated with symptom severity in patients. As such, she attempts to correct some of these abnormalities with molecules that block inflammasome activation. Applying the inflammasome inhibitor MCC950 normalizes inflammasome activity in BD brain organoids and also reduces mtDNA release, indicating a reduction in mitochondrial stress. However, phase II studies of MCC950 for the treatment of autoinflammatory and autoimmune disorders suggest it may cause liver toxicity, so Andreazza also tested a new compound: Bioactive Flavonoid Extract (BFE), which has antioxidant, anti-inflammatory and neuroprotective effects via partial inflammasome inhibition. While inflammasome inhibition was not as strong with BFE, it did reduce mtDNA release to a similar extent. There may be opportunities for elevating the dose or combining it with other treatments to achieve greater impacts.
Altogether Andreazza’s study deconstructs molecular and cellular mechanisms at the intersection of metabolic and immune dysregulation in bipolar disorder. With the novel patient-derived 3D brain organoid
Statement of Significance from Dr. Andreazza:
This study provides a patient-derived, human brain model that links mitochondrial dysfunction, inflammasome activation, and abnormal neuronal activity in bipolar disorder. Led by first author Dana El Soufi El Sabbagh, our team generated cerebral organoids from induced pluripotent stem cells derived from individuals with bipolar disorder and matched controls. The bipolar disorder organoids showed reduced ATP production, altered mitochondrial morphology and membrane potential, increased release of reactive oxygen species and cell-free mitochondrial DNA, and heightened NLRP3 inflammasome sensitivity, accompanied by hyperactive neuronal firing. Importantly, pharmacological inhibition of NLRP3 and a bioactive flavonoid extract partially restored cellular homeostasis, supporting the mitochondria-inflammasome axis as a tractable therapeutic pathway. This work was made possible through a close partnership with Australian collaborators at Deakin University, IMPACT and Barwon Health, who enabled patient recruitment and sample preparation. We are especially grateful to the patients and controls whose participation made this research possible.
A Conversation with Dr. Andreazza:
MitoWorld: Do you have an idea or theory on how mitochondrial dysfunction may play a role in the flip between manic and depressive episodes in bipolar disorder? Are the mitochondria acting differently in these different circumstances?
Dr. Adreazza: Our study did not directly compare cells from manic versus depressive states, so I would be cautious about assigning a specific mitochondrial profile to each episode. What we can say is that mitochondria sit at the centre of processes that are highly relevant to mood-state transitions: ATP production, calcium buffering, oxidative stress, inflammatory signalling, and neuronal excitability. One working model is that an intrinsic mitochondrial vulnerability reduces the energetic flexibility of neurons and astrocytes. During periods of high demand, such as sleep disruption, psychosocial stress, or systemic inflammation, cells may compensate by increasing excitability and stress signalling. This could contribute to manic symptoms in some contexts. Over time, the same system may become energetically depleted, inflammatory pathways may remain activated, and neuronal networks may lose resilience, contributing to depressive symptoms. Rather than mitochondria being entirely different organelles in mania and depression, I think their function may shift dynamically across a spectrum of energetic compensation, oxidative stress, and inflammatory burden.
MitoWorld: Bipolar disorder, and many other psychological conditions where mitochondria are implicated, arise during late adolescence and early adulthood. Do you expect this is when the mitochondria start dysfunctioning, or have they been dysfunctioning all along and reach a certain threshold around this time? Why now?
Dr. Adreazza: I suspect that, in many individuals, mitochondrial vulnerability is present before the first clinical episode, but it may not become functionally limiting until the brain reaches a developmental and environmental threshold. Late adolescence and early adulthood are periods of intense synaptic refinement, circuit maturation, hormonal change, circadian instability, increased psychosocial stress, and, for many people, changes in sleep, diet, and substance exposure. All of these factors place substantial demands on mitochondrial metabolism and redox regulation. If mitochondrial reserve capacity is already reduced, the system may tolerate early development but become less able to adapt when neuronal circuits require more precise energy regulation. Our cerebral organoid data support the idea of an intrinsic, patient-derived cellular vulnerability, because the mitochondrial, inflammatory, and electrophysiological phenotypes emerged in vitro. However, clinical illness likely reflects the interaction between this vulnerability and developmental timing, genetic background, medications, lifestyle, immune activation, and environmental exposures.
MitoWorld: The use of therapeutics to target the inflammasome in your study sounds promising… what are the next steps for potential treatments along those lines? Are there other therapies targeting mitochondria directly that have been tried? What are the challenges to this approach?
Dr. Adreazza: The next step is replication and refinement. We need to test a larger and more clinically diverse set of patient-derived organoids, define dose-response and timing effects, and determine whether inflammasome modulation improves not only inflammatory markers, but also mitochondrial function and neuronal activity. MCC950 was useful experimentally because it is a selective NLRP3 inhibitor, but concerns about hepatotoxicity make it less straightforward as a clinical path. This is why compounds such as the bioactive flavonoid extract are interesting: they may provide broader antioxidant and anti-inflammatory effects, although their potency, active components, pharmacokinetics, safety, and ability to reach the brain need careful evaluation. Mitochondria-directed strategies have also been explored in mood disorders, including agents that influence oxidative stress, bioenergetics, and mitochondrial resilience, such as N-acetylcysteine, coenzyme Q10, creatine, and the mitochondrial effects of lithium. The challenge is that mitochondria are essential in every tissue, and bipolar disorder is biologically heterogeneous. We will need biomarkers to identify who has a mitochondria-inflammatory phenotype and to monitor target engagement.
MitoWorld: How did you become interested in the connection between mitochondria and bipolar disorder? Were you surprised to find this integral relationship?
Dr. Adreazza: My interest developed from the observation that bipolar disorder is not only a disorder of mood regulation, but also a disorder associated with systemic metabolic and inflammatory abnormalities. Over many years, studies from our group and others identified oxidative stress, altered mitochondrial enzymes, and cell-free mitochondrial DNA in blood and brain samples from individuals with bipolar disorder. Those findings made mitochondria a compelling biological link between cellular metabolism, immune activation, and neuronal function. I was not surprised that mitochondria were involved, but I was struck by how clearly the organoid model connected these domains. In the same patient-derived system, Dana El Soufi El Sabbagh and the team could observe impaired mitochondrial energetics, increased mitochondrial stress signals, heightened NLRP3 inflammasome sensitivity, and altered neuronal firing. That integration is important because it moves us beyond isolated biomarkers and toward a mechanistic framework for understanding how cellular stress may influence brain circuit function.
MitoWorld: What is next for this research?
Dr. Adreazza: The immediate priority is to expand the cohort and incorporate clinical information that may explain biological heterogeneity, such as illness stage, predominant polarity, medication exposure, metabolic status, inflammatory burden, and treatment response. We also want to improve the organoid platform by adding greater cellular complexity, including microglia and vascular-like components, because immune and metabolic signalling in the brain depends on interactions among multiple cell types. Longitudinal organoid studies will allow us to follow mitochondrial function, inflammasome activity, and neuronal excitability over developmental time. Another important direction is therapeutic screening. Patient-derived organoids can help us test whether targeting the mitochondria-inflammasome axis restores cellular homeostasis and whether responses differ among individuals. Ultimately, our goal is to integrate organoid biology with blood-based biomarkers, such as cell-free mitochondrial DNA and metabolomic profiles, to move toward more precise, mechanism-based interventions for bipolar disorder.
MitoWorld: Anything else you would like the audience to know?
Dr. Adreazza: I would like to emphasize that this work was highly collaborative. Dana El Soufi El Sabbagh, the first author, led the experimental work across iPSC culture, cerebral organoid generation, mitochondrial assays, inflammatory assays, data analysis, interpretation, and manuscript preparation. The project also depended on an international partnership with our Australian colleagues at Deakin University, IMPACT and Barwon Health, whose expertise in clinical phenotyping, patient selection, and sample preparation made the patient-derived model possible. We are deeply grateful to the individuals living with bipolar disorder, as well as the healthy control participants, who contributed samples and trusted us to use them responsibly. Their participation allows us to study disease mechanisms in a human cellular context that would otherwise be inaccessible. I hope the audience sees this study not as a final answer, but as a platform for building more biologically precise and compassionate approaches to understanding and treating bipolar disorder.
Reference:
iPSC-derived cerebral organoids reveal mitochondrial, inflammatory and neuronal vulnerabilities in bipolar disorder. El Soufi El Sabbagh, D., Kolinski Machado, A., Pappis, L., Beroncal, E. L., Ji, D., Nader, G., Ravi Chander, P., Choi, J., Duong, A., Jeong, H., Panizzutti, B., Bortolasci, C. C., Szatmari, A., Carlen, P., Hahn, M., Attisano, L., Berk, M., Walder, K., & Andreazza, A. C. (2025). Translational Psychiatry, 15, Article 303. https://doi.org/10.1038/s41398-025-03529-7